For Some Representative Metals, Two Possible Ions Can Be Formed. Why?
Chemical bonding, its types, properties, along with chemical reactions is ane of the cornerstones of an interesting science called chemical science. In this article, we will describe all aspects of chemical bonds, their importance in science and much more.
Definition
What is a chemic bail? The chemical bond is the mutual adhesion of atoms in a molecule and the crystal lattice, as a result of the action of the force of attraction that exists between atoms. It is thanks to chemical bonds that various chemical compounds are formed; this is the nature of the chemical bond.
Formation
The bones answer is that atoms are trying to attain the well-nigh stable (lowest-energy) state that they tin can. Many atoms become stable when their valence shell is filled with electrons or when they satisfy the octet dominion (past having eight valence electrons). If atoms don't take this arrangement, they'll "desire" to accomplish it past gaining, losing, or sharing electrons via bonds. Then now you know the answer to the question "what causes a chemical bond to grade betwixt atoms?"
Types
The machinery of the formation of a chemical bail strongly depends on its blazon. In general, there are 4 types of chemical bonds:
- Covalent chemical bail (which in turn can be polar and non-polar)
- Ionic bond
- Hydrogen bond
- Metallic bail
Every bit for the covalent chemical bail, in that location is a divide article on our website, and yous tin can read more in the link. We but notice that the covalent bond is the strongest chemical bond. Further, we will depict in more detail all the other main types of chemic bonds.
Ionic Bond
Ionic bonds course when the common electric attractions of two ions have different charges. In such chemical bonds ions unremarkably are unproblematic, consisting of one atom of the substance.
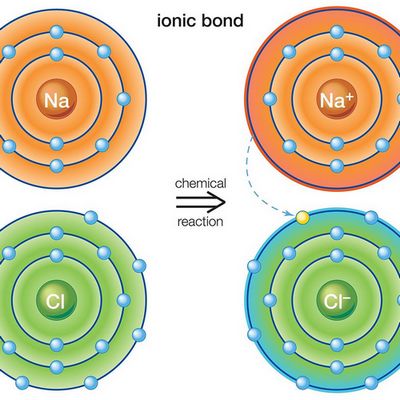
Characteristics properties of ionic bonds are the absence of saturation in information technology. As a issue, a very different amount of oppositely charged ions can join an ion or even a whole group of ions. The ionic bond case is cesium fluoride chemical compound CsF, in which the level of "ionity" is nearly 97%.
Hydrogen Bond
Long earlier the modern theory of chemical bonds was form scientists-chemists noticed that hydrogen compounds with not-metals possess various surprising backdrop. For example, the boiling temperature of the water with hydrogen fluoride is much college than it could be. This is a good case of a hydrogen bond.
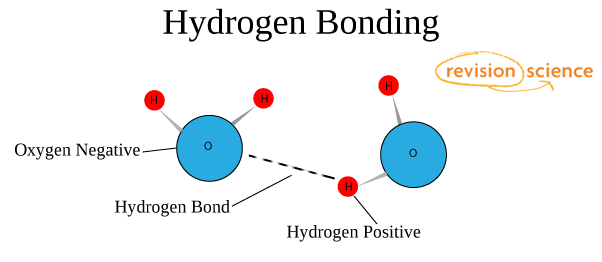
This picture shows the formation of a hydrogen chemic bond.
The nature and properties of the hydrogen bond are due to the power of the hydrogen atom H to grade another chemical bond. The reason for the formation of such a bond is the properties of electrostatic forces. For case, the total electron deject in a molecule of hydrogen fluoride is so biased towards fluorine that the space around the atom of this substance is saturated with a negative electrical field. Around the hydrogen atom, deprived of its but electron, everything is exactly the reverse, and its electron field is much weaker and, as a result, has a positive charge. Positive and negative charges are attracted, so in such a elementary way a hydrogen bond forms.
Metal Bond
Metals accept their own type of chemical bond – the atoms of all metals are located in a sure way. The gild of their arrangement is called a crystal lattice. The electrons of different atoms course a mutual electron cloud, while they weakly interact with each other.
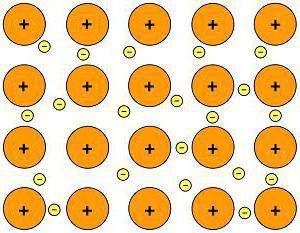
This is a metallic bond.
If you meet a cleaved fence information technology could hateful that metallic bonds are cleaved. Yet this is not the problem considering yous tin can order metal debate repair at quickfencerepair.com.
How to Determine the Blazon of Bond?
The blazon of chemical bond depends on the substances taking part in it. If the substances are metallic and not-metal, then the bail between them is ionic. If two substances are metallic, then the bond betwixt them is metallic. If two substances are non-metal, then the bond is covalent.
Backdrop
Different quantitative characteristics are used to compare different chemical reactions such as:
- length
- energy,
- polarity
- the social club.
Allow united states depict them in more detail.
The bail length is the equilibrium distance betwixt the nuclei of atoms that are connected past a chemical bond.
The energy of a chemic bond determines its force. In this case, the energy means the effort required to interruption the chemic bond and to separate the atoms.
The polarity of a chemical bond shows how much the electron density is shifted to i of the atoms. The ability of atoms to shift their electron density towards themselves or, in unproblematic terms, "pull a blanket over themselves" is chosen electronegativity in chemistry.
The guild of a chemical bond (in other words, the multiplicity of a chemical bond) is the number of electron pairs entering into a chemical bond. The guild can be both integer and fractional, the higher it is, the greater the number of electrons carries out chemical bonding and the more hard information technology is to break it.
References and Further Reading
- Rioux, F. (2001). "The Covalent Bond in H2". The Chemical Educator. 6 (v): 288–290. doi:10.1007/s00897010509a.
- Lewis, Gilbert North. (1916). "The Atom and the Molecule". Journal of the American Chemical Guild. 38 (4): 772. doi:10.1021/ja02261a002. a copy
- Бор Н. (1970). Избранные научные труды (статьи 1909–1925). 1. М.: «Наука». p. 133.
- Svidzinsky, Anatoly A.; Marlan O. Scully; Dudley R. Herschbach (2005). "Bohr's 1913 molecular model revisited". Proceedings of the National Academy of Sciences. 102 (34[1]): 11985–11988. arXiv:physics/0508161. Bibcode:2005PNAS..10211985S. doi:10.1073/pnas.0505778102. PMC 1186029. PMID 16103360.
- Laidler, Grand. J. (1993). The World of Concrete Chemistry. Oxford University Press. p. 346. ISBN 978-0-19-855919-ane.
- James, H.H.; Coolidge, A S. (1933). "The Footing State of the Hydrogen Molecule". Journal of Chemical Physics. 1 (12): 825–835. Bibcode:1933JChPh…1..825J. doi:10.1063/1.1749252.
- "Bond Energies". Chemical science Libre Texts. Retrieved 2019-02-25.
- Atkins, Peter; Loretta Jones (1997). Chemistry: Molecules, Matter and Alter. New York: W.H. Freeman & Co. pp. 294–295. ISBN 978-0-7167-3107-8.
Video

Author: Pavlo Chaika, Editor-in-Main of the journal Poznavayka
When writing this article, I tried to brand information technology as interesting and useful every bit possible. I would exist grateful for any feedback and constructive criticism in the form of comments to the article. Y'all tin also write your wish/question/proposition to my mail pavelchaika1983@gmail.com or to Facebook.
Source: https://www.poznavayka.org/en/chemistry/chemical-bonding/
0 Response to "For Some Representative Metals, Two Possible Ions Can Be Formed. Why?"
Post a Comment